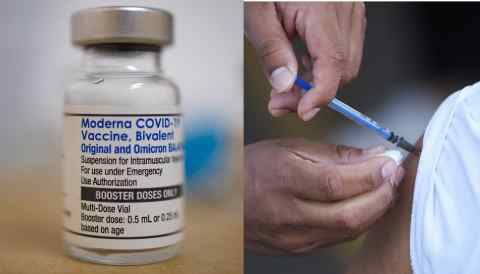
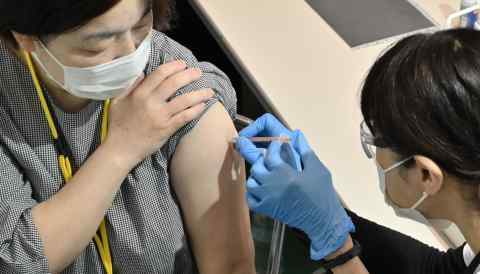
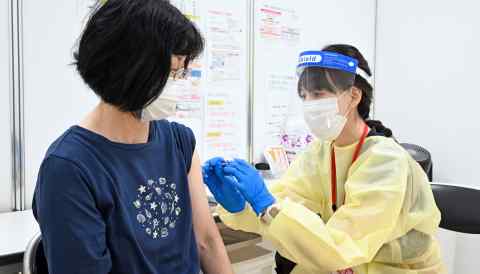
TOKYO -- Japan will consider making vaccines and medical treatments that have yet to be domestically approved available in an emergency after regulatory roadblocks delayed its COVID-19 vaccination campaign as cases spiked.
Amid criticism over Japan's slow pace of inoculation, Prime Minister Yoshihide Suga hopes to update existing legislation. If approved, new vaccines and treatments that are already in use overseas would temporarily be made available to the public before a clinical trial can be completed in Japan.